G4 Binders As Potent Selective Transcription Inhibitors
Qualigen in-licensed the G4 selective transcription inhibitor platform from University College London (UCL) in 2022. The licensed technology comprises lead compound QN-302 and backup compounds that target regulatory regions of cancer genes that down-regulate gene expression in multiple cancer pathways. Developed by Dr. Stephen Neidle and his research group at UCL, the G4 binding concept is derived from 30+ years of nucleic acid research, including G4s, which are higher-order DNA and RNA structures formed by sequences containing guanine-rich repeats. G4s are overrepresented in telomeres and promoter sequences and untranslated regions of many oncogenes. Therefore, their prevalence is significantly greater in cancer cells compared to normal human cells1.
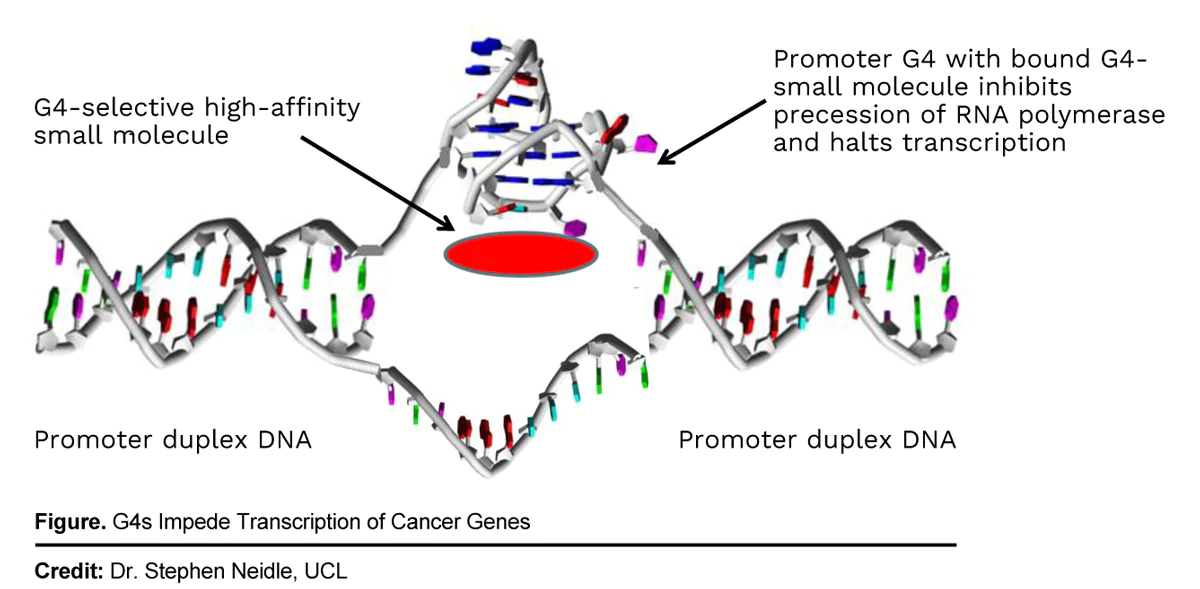
Bioinformatics can predict how G4-selective molecules such as QN-302 target the regulatory regions of cancer genes. Stable G4-QN-302 complexes can impede the replication, transcription, and translation of those cancer genes containing G4s, as the drug’s binding to G4s stabilizes them against possible “unwinding.” This leads to cancer cell death through apoptosis. As a result, G4 binders like QN-302 could be efficacious in treating various cancer types that have a high prevalence of G4s. Qualigen plans to initiate a Phase I, multicenter, open-label, dose-escalation and dose expansion, safety, pharmacodynamic and pharmacokinetic study of intravenous QN-302 in patients with advanced or metastatic solid tumors in the second half of 2023. The Company anticipates the dosing of at least 24 patients in the Phase 1 trial will be completed by end 2024, funded in part by proceeds received by the divestiture of the Company’s diagnostics business in July 2023.
High Affinity G4 Binder QN-302 as Treatment for Pancreatic Cancer and Other Solid Tumors
Pancreatic cancer is the tenth most common cancer type for incidence but the fourth most common type for deaths in the United States and has one of the lowest survival rates of all cancer types. The chemotherapeutic drug gemcitabine has been standard of care for patients with metastatic pancreatic cancer for more than 15 years. Numerous clinical trials have tested new drugs either alone or in combination with gemcitabine2 but with relatively little success. QN-302 is potentially a first-in-class small molecule drug candidate with significant efficacy and activity against pancreatic cancer and other G4 prevalent tumors as compared to existing agents, with a distinct mechanism of action and promising preclinical target profile.
In-vitro and in-vivo studies have shown that G4 stabilization by QN-302 resulted in inhibition of target gene expression and cessation of cell growth in various cancers, including pancreatic ductal adenocarcinoma (“PDAC”) which represents the majority of pancreatic cancers. In in vitro studies, QN-302 potently inhibited the growth of several PDAC cell lines at low nanomolar concentrations. Likewise, in vivo QN-302 showed a longer survival duration in a KPC genetic mouse model for pancreatic cancer than gemcitabine has historically shown. Additional preclinical in vivo studies suggest activity in gemcitabine-resistant PDAC. Data further demonstrated that QN-302 had significant anti-tumor activity in three patient-derived PDAC patient-derived xenograft models. Early safety indicators suggest no adverse toxic effects at proposed therapeutic doses in pancreatic cancer mouse in-vivo models. In addition to safety and efficacy studies, there have been transcriptome analyses of oncogenes to evaluate potential biomarkers associated with response in G4-prevalent tissues. One such gene being examined is S100P, commonly found in in PDAC.
Potential Advantages
- 1. Orphan Drug Designation awarded by FDA for pancreatic cancer
- Composition of matter IP protection covering QN-302 and back-up compounds
- QN-302 is a small molecule with straightforward four-step synthesis
- Evidence for high affinity G4 target engagement
- Nanomolar anti-proliferative activity in variety of cancer cell lines, including MIA-PACA2 cells
- Anti-tumor activity in-vivo in multiple pancreatic cancer models, including PDx models
- Long half-lives (>24hrs) and favorable in vivo exposure
- 8. No adverse toxic effects in-vivo at proposed therapeutic doses in murine models
- No significant adverse effects on blood chemistry in pancreatic cancer in-vivo model
- 10. Promising in-vivo activity against hormone-resistant prostate cancer and compelling in vitro activity in GIST (Gastrointestinal Stroma Tumor) cell lines
- 11. Potential for tumor-agnostic, G4-biomarker based clinical approach and additional Orphan Drug designations
Please contact [email protected] for more information.
Posters & Publications
- Poster – AACR Special Conference, September 2023
The Pan G-Quadruplex experimental drug QN-302 in PDAC: identification of potential biomarkers for clinical studies - Poster – AACR Special Conference, September 2023
Target genes in pancreatic cancer cells of the Pan G-Quadruplex clinical candidate compound QN-302 revealed by comparative transcriptome profiling - Poster – AACR Annual Meeting, April 2023
A comparison of the activity of the quadruplex-targeting experimental drugs QN-302 and CX-5461 (Pidnarulex) in wild-type and gemcitabine-resistant pancreatic cancer cells - Poster – AACR Annual Meeting, April 2023
The potent quadruplex-binding compound QN-302 down-regulates the S100P gene in in vitro and in vivo models of pancreatic cancer: a potential therapeutic target and biomarker for PDAC - Poster – AACR Annual Meeting, April 2023
The potent quadruplex-binding compound QN-302 shows anti-tumor activity as a monotherapy in an orthotopic in vivo model of pancreatic cancer - Poster – AACR Annual Meeting, April 2023
Structure-based design rules for potent quadruplex-binding compounds based on the naphthalene diimide core - Research Article – Molecules, March 2023
The Potent G-Quadruplex-Binding Compound QN-302 Downregulates S100P Gene Expression in Cells and in an In Vivo Model of Pancreatic Cancer - Poster — AACR Special Conference, September 2022
The quadruplex-binding compound QN-302 in the MIA-PaCa2 pancreatic adenocarcinoma model shows no cardiac or neurological liabilities at therapeutic doses (Authors: Ahmed Ahmed, Tariq Arshad, and Stephen Neidle) - Poster — AACR Special Conference, September 2022
The quadruplex-binding compound QN-302 targets the S100P gene in PDAC (Authors: Ahmed Ahmed, William Greenhalf, Tariq Arshad, and Stephen Neidle) - Poster — AACR Annual Meeting, April 2022
The potent quadruplex-binding compound QN-302 shows potent anti-proliferative activity in a prostate cancer cell panel and anti-tumor activity in an in vivo model of metastatic prostate cancer (Authors: Nicole Williams, Jenny Worthington, Stephen Neidle, Ahmed Ahmed) - Poster — AACR Annual Meeting, April 2022
The potent quadruplex-binding compound QN-302 shows anti-tumor activity in patient-derived in vivo models of pancreatic cancer (Authors: Stephen Neidle, Ahmed Ahmed, Richard Angell, Sally Oxenford) - Poster — AACR Annual Meeting, April 2022
Structure-based design of quadruplex-binding small molecule compounds: the essential role of water molecules (Author: Stephen Neidle) - Book — Elsevier Inc. (elsevier.com), October 2021
Principles of Nucleic Acid Structure (Authors: Stephen Neidle, Mark Sanderson) - Research Article — Journal of Biological Chemistry (jcb.org), January 2021
Beyond the double helix: DNA structural diversity and the PDB - Research Article — Nucleic Acids Research (academic.oup.com), January 2021
Water spines and networks in G-quadruplex structures - Research Article — ACS Medicinal Chemistry Letters (acs.org), January 2021
Asymmetrically Substituted Quadruplex-Binding Naphthalene Diimide Showing Potent Activity in Pancreatic Cancer Models - Book — Elsevier Inc. (elsevier.com), August 2020
Quadruplex Nucleic Acids As Targets For Medicinal Chemistry - Research Article — Scientific Reports (nature.com), July 2020
A G-quadruplex-binding compound shows potent activity in human gemcitabine-resistant pancreatic cancer cells - Research Article — Molecules (mdpi.com), July 2020
A G-Quadruplex-Binding Small Molecule and the HDAC Inhibitor SAHA (Vorinostat) Act Synergistically in Gemcitabine-Sensitive and Resistant Pancreatic Cancer Cells
References
- Hänsel-Hertsch R et al. G-quadruplex structures mark human regulatory chromatin. Nat Genet. 2016 Oct;48(10):1267-72. doi: 10.1038/ng.3662. Epub 2016 Sep 12. PMID: 27618450.
- “Combination of Nab-Paclitaxel and Gemcitabine Improves Survival in Patients with Metastatic Pancreatic Cancer.” www.cancer.gov/types/pancreatic/research/nab-paclitaxel-gemcitabine
- Ahmed A et al. Asymmetrically Substituted Quadruplex-Binding Naphthalene Diimide Showing Potent Activity in Pancreatic Cancer Models. ACS Med Chem Lett. 2020 Jul 16;11(8):1634-1644. doi: 10.1021/acsmedchemlett.0c00317
- Ahmed et al. A G-quadruplex-binding compound shows potent activity in human gemcitabine-resistant pancreatic cancer cells. Sci Rep. 2020 Jul 22;10(1):12192. doi: 10.1038/s41598-020-68944-w.